CAR-T Cell Therapy
- 17 Apr 2026
In News:
In a landmark medical achievement reported in early 2026, scientists successfully utilized Chimeric Antigen Receptor (CAR) T-cell therapy to treat a patient suffering from three concurrent, life-threatening autoimmune diseases. This patient, who had remained resistant to traditional treatments for years, entered remission, marking a paradigm shift in how "living drugs" can be used beyond oncology.
What is CAR-T Cell Therapy?
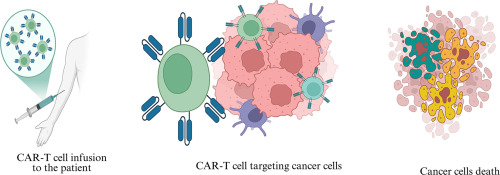
CAR-T cell therapy is an advanced form of immunotherapy and gene therapy that re-engineers a patient’s own immune system to target specific diseased cells.
- The Component Cells: It focuses on T-lymphocytes (T cells), a type of white blood cell responsible for identifying and destroying foreign pathogens and abnormal cells.
- The "Chimeric" Aspect: In a laboratory, these T cells are genetically modified to produce synthetic proteins called Chimeric Antigen Receptors (CARs). These receptors act like "navigation systems," allowing T cells to recognize and bind to specific antigens (proteins) on the surface of target cells.
Mechanism of Action: The Step-by-Step Process
The production and administration of CAR-T cells involve a highly sophisticated multi-step process:
- Leukapheresis: T cells are extracted from the patient’s blood through a specialized filtering process.
- Genetic Engineering: Using viral vectors, the gene for the synthetic CAR receptor is inserted into the T cells.
- Expansion: The modified CAR-T cells are grown in large quantities in a laboratory setting.
- Infusion: The "supercharged" cells are infused back into the patient’s bloodstream.
- Targeted Destruction: The CAR-T cells identify the target antigens, bind to them, and trigger a lethal immune response against the diseased cells. Unlike traditional drugs, these cells can continue to multiply in the body, providing long-term surveillance.
Expanding Horizons: From Oncology to Autoimmunity
Traditionally, CAR-T therapy has been the last line of defense for specific B-cell malignancies (blood cancers), including:
- B-cell Acute Lymphoblastic Leukemia (ALL)
- Multiple Myeloma
- Various types of Lymphoma (Follicular, Mantle cell, and Diffuse large B-cell).
The 2026 Breakthrough:
Autoimmune diseases occur when the immune system mistakenly attacks healthy tissues. By programming CAR-T cells to eliminate the specific B-cells producing "auto-antibodies," doctors have successfully induced remission in patients with multiple systemic autoimmune conditions simultaneously. This offers hope for treating diseases like Lupus, Scleroderma, and Myositis where standard immunosuppressants fail.
Challenges and Adverse Effects
Despite its revolutionary potential, CAR-T therapy is associated with significant clinical risks that require intensive monitoring:
- Cytokine Release Syndrome (CRS): A severe immune overreaction leading to hyperinflammation, high fever, and potential organ damage. Clinical data indicates this occurs in approximately 12% of cases.
- Hematological Toxicity: * Neutropenia: Low neutrophil count (seen in 96% of participants), which drastically increases the risk of life-threatening infections.
- Thrombocytopenia: Low platelet count (reported in 65% of patients), raising the risk of internal bleeding.
- Anemia: Low red blood cell count (reported in 61% of participants), causing extreme fatigue.
- Cost and Accessibility: As a personalized "living drug," the therapy remains prohibitively expensive and requires specialized infrastructure found only in advanced tertiary care centers.
Significance for India
India has recently entered the CAR-T space with the indigenous NexCAR19 (developed by IIT Bombay and Tata Memorial Hospital). The expansion of this technology into autoimmune treatment is significant for India’s healthcare landscape, as it could eventually provide a one-time "cure" for chronic conditions that currently require lifelong, expensive medication.
